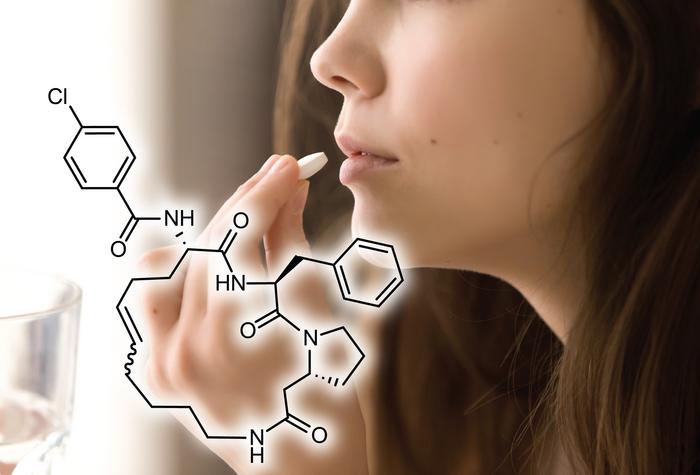
Cyclic peptides demonstrate superior stability compared to linear counterparts through structural constraints that eliminate the flexibility of vulnerable terminal residues. The closed-loop architecture removes exposed N-terminal and C-terminal groups that exopeptidases typically attack. Resources through bluumpeptides.com catalog stability data comparing cyclic and linear peptide variants across enzymatic and thermal challenges. Backbone cyclization creates a continuous peptide bond chain without free ends. Cyclic peptides maintain biological activity longer than linear sequences. They withstand harsher conditions during storage and formulation.
Protease resistance mechanisms
- Linear peptides are sequentially degraded by exopeptidases by cleaving free amino acids on their ends
- A serine aminopeptidase breaks down amino acids one by one, whereas a carboxypeptidase attacks C-termini, leading to bidirectional degradation pathways.
- Due to the elimination of accessible termini, cyclization prevents activation of exopeptidases due to their requirement for free amino or carboxyl groups
- Protease active sites evolved recognizing extended linear substrates, making cyclic structures geometrically incompatible with many enzyme binding pockets, reducing cleavage susceptibility substantially
Linear peptides present easy targets. Proteases evolved to degrade dietary proteins. These enzymes efficiently break down ingested peptides. The digestive tract contains numerous proteases. Aminopeptidases start at the N-termini. Carboxypeptidases begin at the C-termini. Between them, linear peptides get dismantled rapidly. Half-lives in serum measure minutes or hours. Cyclic peptides change this equation. No free termini exist. Exopeptidases find nothing to grab. They need that initial amino acid. Without it, catalysis cannot start. The peptide circulates intact much longer. Serum half-lives extend to hours or days. Some cyclic peptides persist for weeks.
End opeptidases pose different challenges. They cut internal bonds. Cyclization alone does not prevent this. But conformational restriction helps. Rigid cyclic structures adopt specific shapes. Many protease recognition sites require extended conformations. The cyclic peptide cannot extend properly. The protease active site cannot accommodate the constrained structure. Cleavage fails or occurs much more slowly.
Conformational entropy reduction
Cyclization dramatically decreases conformational flexibility. Linear peptides sample many structures. Rotation around single bonds permits countless configurations. Each conformation exists temporarily. The ensemble averages across all possibilities. This flexibility carries an entropic cost. Bioactive conformations represent small fractions of the ensemble. Most conformations lack activity. When a linear peptide binds its target, it must adopt the correct shape. The entropic penalty for losing flexibility reduces binding affinity. Cyclic peptides pay this entropy cost during synthesis instead. The bioactive conformation gets locked in.
Reduced sampling improves binding. Cyclic peptides exist predominantly in active conformations. Target binding requires minimal conformational adjustment. Association rates increase. Dissociation rates decrease. Both effects strengthen interactions. Potency often improves compared to linear versions. Ring size determines rigidity. Small rings constrain more than large ones. Three to seven amino acid cycles show extreme rigidity. Larger rings retain some flexibility. The optimal size balances rigidity against synthesizing the bioactive conformation. Too much constraint in the wrong shape destroys activity.
Bioavailability enhancement
Proteolytic stability directly improves bioavailability. Peptides administered orally face gastric and intestinal proteases. Linear therapeutic peptides rarely survive. Injection bypasses the gut, but serum proteases still attack. Cyclic peptides resist both environments better. More intact drug reaches targets. Some cyclic peptides cross membranes more efficiently than linear forms. Passive diffusion requires proper lipophilicity and hydrogen bonding characteristics. Cyclization can optimize both properties. Intramolecular hydrogen bonds reduce the number interacting with water. This decreases polar surface area. Membrane permeability increases.
Transporter compatibility varies. Some cyclic peptides engage uptake transporters. Others evade efflux pumps. Scale-up proceeds smoothly. Cyclic peptides crystallize well. Good crystallinity facilitates purification. Regulatory agencies approve cyclic peptides. Several reached the market. More advances through clinical trials. Commercial viability no longer questions cyclic peptide drugs.